Biomolecules | Free Full-Text | BabyBoom: 3-Dimensional Structure-Based Ligand and Protein Interaction Prediction by Molecular Docking

Describing static correlation in bond dissociation by Kohn–Sham density functional theory: The Journal of Chemical Physics: Vol 122, No 9

Collision Induced Dissociation of Deprotonated Isoxazole and 3-Methyl Isoxazole via Direct Chemical Dynamics Simulations | Journal of the American Society for Mass Spectrometry

PDF) The impact of the self-interaction error on the density functional theory description of dissociating radical cations: Ionic and covalent dissociation limits

Adsorption and Dissociation of Molecular Hydrogen on Palladium Clusters Supported on Graphene | The Journal of Physical Chemistry C

Stable Surfaces That Bind Too Tightly: Can Range-Separated Hybrids or DFT+U Improve Paradoxical Descriptions of Surface Chemistry? - Abstract - Europe PMC

Oxidative Rearrangements of Tricyclic Vinylcyclobutane Derivatives - Grota - 2006 - Chemistry – A European Journal - Wiley Online Library

Faster Dissociation: Measured Rates and Computed Effects on Barriers in Aryl Halide Radical Anions | Journal of the American Chemical Society

Modeling the Charge Transfer between Alkali Metals and Polycyclic Aromatic Hydrocarbons Using Electronic Structure Methods | The Journal of Physical Chemistry A
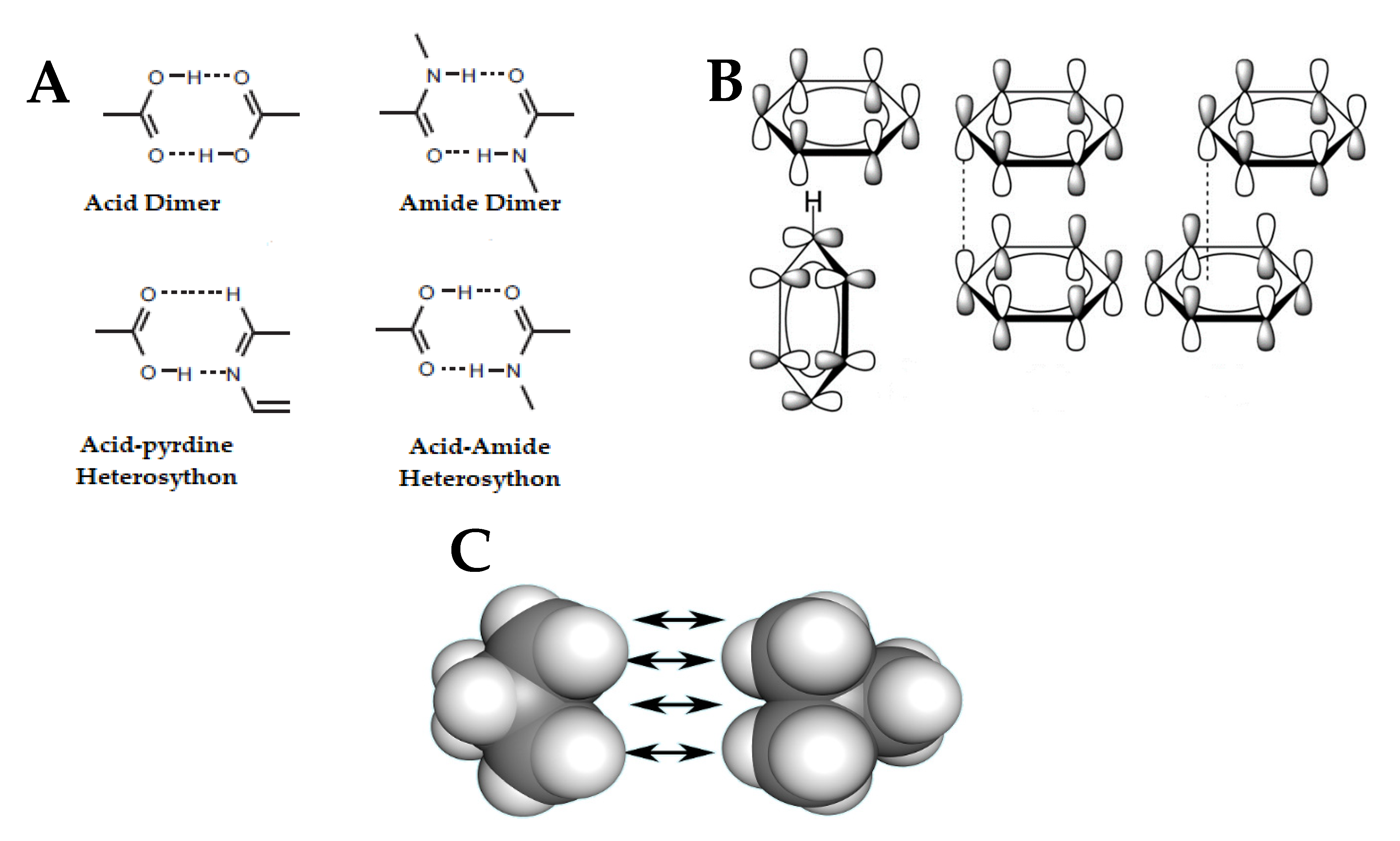
Crystals | Free Full-Text | Nano- and Crystal Engineering Approaches in the Development of Therapeutic Agents for Neoplastic Diseases

Oxidative Rearrangements of Tricyclic Vinylcyclobutane Derivatives - Grota - 2006 - Chemistry – A European Journal - Wiley Online Library

Adsorption and Dissociation of Molecular Hydrogen on Palladium Clusters Supported on Graphene | The Journal of Physical Chemistry C

Adsorption and Dissociation of Molecular Hydrogen on Palladium Clusters Supported on Graphene | The Journal of Physical Chemistry C

Adsorption and Dissociation of Molecular Hydrogen on Palladium Clusters Supported on Graphene | The Journal of Physical Chemistry C

Adsorption and Dissociation of Molecular Hydrogen on Palladium Clusters Supported on Graphene | The Journal of Physical Chemistry C
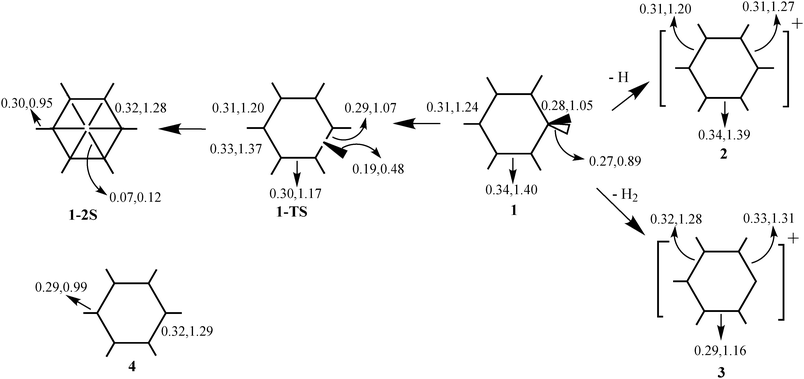
Energetic and electron density analysis of hydrogen dissociation of protonated benzene - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/B906961E
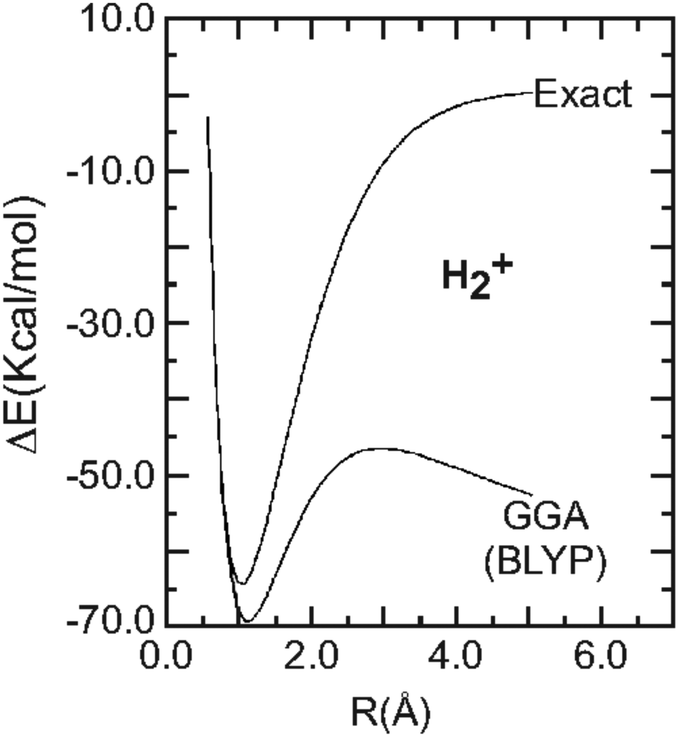
Piecewise linearity, freedom from self-interaction, and a Coulomb asymptotic potential: three related yet inequivalent properties of the exact density ... - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/D0CP02564J

Investigation of adsorption, dissociation, and diffusion properties of hydrogen on the V (1 0 0) surface and in the bulk: A first-principles calculation - ScienceDirect

PDF) Spin-component-scaled and dispersion-corrected second-order Møller-Plesset perturbation theory: A path toward chemical accuracy